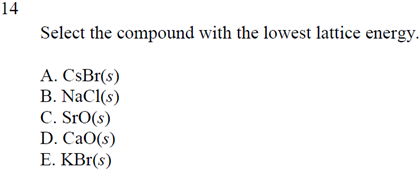
"Crystal-field induced dipoles in heteropolar crystals I: Concept". Advanced Inorganic Chemistry (2d Edn.) New York:Wiley-Interscience. The difference in lattice energies between MgCl2 and CaCl2 is 269 kJ, but the difference between. Brown, The chemical Bond in Inorganic Chemistry, IUCr monographs in crystallography, Oxford University Press, 2002, ISBN 0-19-850870-0 Potential energy decreases with separation of the charges. Boca Raton, FL: CRC Press, Taylor & Francis Group. Ionisation: Ca A ( g) Ca A 2 + A ( g) + 2 e A H H 2 0 + 2422 kJ mol 1. Sublimation: (g) Ca A ( s) Ca (g) H H 1 0 + 121 kJ mol 1. Ca A ( s) + Cl A 2 A ( g) CaCl A 2 A ( s) H f H f 0 795 kJ mol 1. CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data. Calculate the lattice energy of CaCl2 from the given data.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
